Study Finds Record High Levels of Toxic Fire Retardants in Breast Milk from American Mothers
In the first nationwide tests for chemical fire retardants in the breast milk of American women, the Environmental Working Group (EWG) found unexpectedly high levels of these little-known neurotoxic chemicals in every participant tested.
The average level of bromine-based fire retardants in the milk of 20 first-time mothers was 75 times the average found in recent European studies. Milk from two study participants contained the highest levels of fire retardants ever reported in the United States, and milk from several of the mothers in EWG's study had among the highest levels of these chemicals yet detected worldwide.
These results confirm recently published findings from University of Texas researchers, as well as other U.S. studies, that American babies are exposed to far higher amounts of fire retardants than babies in Europe, where some of these chemicals have already been banned. In the United States, only California and Maine have acted to restrict the use of these chemicals.
Breast milk is bestEven women with very high levels of fire retardants in their breast milk should continue to breastfeed their babies. There are two main reasons why. First, adverse effects on learning and behavior are strongly associated with fetal exposure to persistent pollutants, not with breast milk exposure. And second, breastfeeding appears to overcome some of the harmful effects of high fetal exposure to persistent chemicals. Breast milk data are very useful, however, because they are an excellent measure of fetal blood levels, and fetal exposure to fire retardants. |
Like PCBs, their long-banned chemical relatives, brominated fire retardants are persistent in the environment and bioaccumulative, building up in people’s bodies over a lifetime. Brominated fire retardants impair attention, learning, memory, and behavior in laboratory animals at surprisingly low levels. The most sensitive time for toxic effects is during periods of rapid brain development. Fire retardants in breast milk are one measure of the chemicals that a mother passes on not only to her nursing infant, but more importantly, to the unborn fetus, which is most vulnerable to impacts from neurotoxic chemicals.
Brominated fire retardants are in hundreds of everyday products, including furniture, computers, TV sets and automobiles. Studies worldwide have found them to be building up rapidly in people, animals and the environment, where they persist for decades. Research on animals shows that fetal exposure to minute doses of brominated fire retardants at critical points in development can cause deficits in sensory and motor skills, learning, memory and hearing. Levels of particularly toxic and bioaccumulative types of brominated fire retardants, known as polybrominated diphenyl ethers (PBDEs), are by far highest in the United States and Canada compared to levels in any other country. Together, the US and Canada account for almost half of global PBDE use.
Chemical fire retardants are not necessary for fire safety: Some manufacturers, from furniture makers to computer companies, have achieved the same level of safety by redesigning their products to be inherently less flammable without chemical treatments. The European Union has banned the most toxic forms of PBDEs beginning next year, and some Asian countries are close behind. But the U.S. Environmental Protection Agency has set no safety standards or other regulations for their manufacture, use or disposal. Only one state, California, has banned some chemical fire retardants, with the phase-out to be complete by 2008. By then, if fire retardants continue to be used at the present level, another 365 million pounds of these toxicants will be in Americans' homes, schools, offices, and bodies.
[Download larger version of map in PDF format]
Most brominated fire retardants used in North America are made by two companies, Great Lakes Chemical Corp. of West Lafayette, IN, and Albemarle Corp. of Richmond, VA. While preparing to comply with the EU ban, these companies are spending millions of dollars in Congress and state legislatures lobbying against domestic protections — even working against consumers' right to know what they're buying so they can choose PBDE-free products. Through their dominance of the world market in brominated chemicals, the two corporations are hindering the efforts of other companies to provide customers with safer alternatives.
These results confirm the need for prompt action to reduce American children’s exposures to toxic fire retardants.
- The EPA should phase out all of these toxic fire retardants as quickly as possible. In the interim, all products containing PBDEs should be labeled so that consumers have the option of choosing products without them.
- EPA must screen new and existing chemicals for their health effects. In particular, potential replacement fire retardants must be adequately tested to ensure that they are not persistent, bioaccumulative or toxic. Testing must include the outcomes most relevant to children’s health. Changes in product design that decrease the need for chemical fire retardants should be encouraged over simply switching to different, less studied chemicals.
- The Centers for Disease Control and Prevention should expand its fledgling national biomonitoring program to include a greater number of chemicals and people. The study provides critical data in identifying chemicals that are accumulating in our bodies and in the environment; tracking trends in exposure; providing data needed to more fully understand human health risks; and helping EPA and other agencies effectively transition businesses to safer, less persistent chemicals than those in current common use.
- Congress should increase funding for urgently needed research on toxic fire retardants, including their health effects, how they get into the human body, and current levels of accumulation in people, animals and the environment.
The next PCBs?
As less combustible products like cotton and wood have been replaced by highly flammable synthetic materials in consumer products, chemical fire retardants have become ubiquitous. There are many different kinds of fire retardants with varying degrees of toxicity. A group of brominated fire retardants (BFRs) called polybrominated diphenyl ethers, or PBDEs, have come to the attention of scientists and regulators because of evidence of their environmental persistence and bioaccumulation in living organisms, as well as their toxic effects. Today PBDEs are in thousands of products, in which they typically comprise five to 30 percent of product weight.[1]
During manufacturing, PBDEs are simply mixed into the plastic or foam product, rather than being chemically bound to the material as some other fire retardants are, making them more likely to leach out into the human environment.
Brominated Fire Retardants are found in everyday consumer products
|
Materials used in |
Types of PBDEs used |
Examples of consumer products |
|
Plastics |
Deca, Octa, Penta |
Computers, televisions, hair dryers, curling irons, copy machines, fax machines, printers, coffee makers, plastic automotive parts, lighting panels, PVC wire and cables, electrical connectors, fuses, housings, boxes and switches, lamp sockets, waste-water pipes, underground junction boxes, circuit boards, smoke detectors |
|
Textiles |
Deca, Penta |
Back coatings and impregnation of home and office furniture, industrial drapes, carpets, automotive seating, aircraft and train seating |
|
Polyurethane foam |
Penta |
Home and office furniture (couches and chairs, carpet padding, mattresses and mattress pads) automobile, bus, plane and train seating, sound insulation panels, imitation wood, packaging materials |
|
Rubber |
Deca, Penta |
Conveyor belts, foamed pipes for insulation, rubber cables |
|
Paints and lacquers |
Deca, Penta |
Marine and industry protective lacquers and paints |
Source: [1], [112]
PBDEs are the chemical cousins of PCBs, another family of highly persistent and bioaccumulative toxicants that came to the attention of health officials only after millions of pounds had been released into the environment. In the 26 years since PCBs were banned, numerous studies have documented permanent, neurological impairment to children from low level PCB exposure.[2, 3, 4, 5, 6]
Recent evidence suggests PBDEs and PCBs may work together to cause adverse health effects. Not only do PBDEs appear to be acting through the same pathways as PCBs, but also exposure to a combination of PCBs and PBDEs appears to affect motor skills of lab animals ten times more strongly than exposure to either chemical alone. [7, 8]
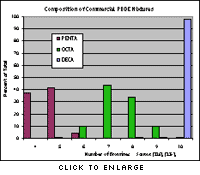
Types of PBDEsThere are 209 types, or congeners, of PBDEs, classified by the number of bromine atoms in a molecule of the chemical: Penta-BDEs have five bromine atoms, octa-BDEs have eight, deca-BDE has 10, and so on. Commercial fire retardants containing PBDEs are actually mixtures of several different congeners, with the three major commercial mixtures called Deca, Penta, and Octa. The common name of the commercial product can be somewhat misleading; the Penta product, for example, is actually a mixture of 40 percent tetra-BDE, 45 percent penta-BDE and 6 percent hexa-BDE congeners. The product known as Deca is mostly made up of PBDEs with 10 bromines, but PBDEs with eight or nine bromines make up about 2% of the mixture. Overall, a small number of PBDE congeners are predominant in commercial fire retardant mixtures and in the environment. Graphic: Composition of Commercial Mixtures |
The use of these fire retardants has skyrocketed in the last three decades, with Penta production almost doubling between 1992 and 2001. [9, 10] Worldwide, Deca is the most widely used of the PBDEs with 83 percent of the global market by weight, followed by Penta with 11 percent and Octa with 6 percent. [10] The market for PBDEs took off after the 1978 ban of a related class of brominated fire retardants called polybrominated biphenyls, or PBBs. Once widely used as fire retardants, PBBs were banned following the detection of contaminated cattle feed in Michigan during 1973 and 1974 that exposed 9 million people to tainted meat and dairy products. [11]
Today, half of the PBDEs used worldwide are used in the Americas — 73 million pounds in 2001 alone. [10] Led by the U.S. and Canada, the Americas consume 95 percent of the global supply of Penta, which is the type most easily taken up by animals and people. An unknown amount of PBDEs, probably millions of pounds, is also imported into the country each year in manufactured goods. Chemical industry analysts say the North American market for brominated fire retardants is $1 billion a year and growing by about 3.7% annually; the European market is a little more than half that size. [12] PCB use in the US peaked at 86 million pounds per year in 1970. PBDE use in the US and Canada is approaching this level with 73 million pounds consumed in 2001. In total, almost as many pounds of PBDEs have been released into the environment in the US as PCBs.
Only eight companies manufacture PBDEs worldwide, with the two largest in the U.S.: Great Lakes Chemical Corp. of West Lafayette, IN, and Albemarle Corp. of Richmond, VA. In 2002, Great Lakes reported total sales for all products of $1.4 billion, up 4% from the previous year. Albermarle reported sales of $980 million, up 7%. [13, 14] The corporations are already notorious as the manufacturers of methyl bromide, a volatile, acutely toxic, ozone-depleting pesticide gas used to fumigate strawberries, tomatoes and other crops. (Albemarle also has the dubious distinction of being a spin-off of Ethyl Corp., whose leaded gasoline additive was banned in the U.S. in 1972.) The main areas of bromine production in the world are southeastern Arkansas, where Great Lakes and Albemarle pump it from underground pools of brine, and Israel, where a company named Dead Sea Bromine extracts it from the briny inland sea. A chemical industry journal describes the global trade in brominated chemicals as "an oligopoly controlled by Albemarle, Great Lakes and the Dead Sea Bromine Group." [15]
Brominated fire retardants widely used in the Americas
Global Use of PBDEs in 2001 (pounds)
|
Commerical PBDE Product |
Americas |
Europe |
Asia |
Other |
Percent used in the Americas |
|
Deca |
54,010,000 |
16,760,000 |
50,710,000 |
2,315,000 |
44% |
|
Penta |
15,650,000 |
331,000 |
331,000 |
221,000 |
95% |
|
Octa |
3,307,000 |
1,345,000 |
3,307,000 |
397,000 |
40% |
Source: [10]
Despite their heavy use, until recently data were scarce on the toxicity or environmental fate of PBDEs. But in the last few years, it has become clear that PBDEs and other brominated fire retardants have joined PCBs, DDT and dioxin on the list of persistent, bioaccumulative chemicals contaminating people, animals and the environment everywhere on the planet. These fire retardants are now found in house dust, sewage sludge and the water and sediments of rivers, estuaries and oceans. They've been found in the tissues of whales, seals, birds and bird eggs, moose, reindeer, mussels and dozens of species of freshwater and marine fish. [16, 17, 18, 19, 20, 21] Brominated fire retardants have been detected in birds and marine mammals in remote locations including the North Sea, the Baltic Sea and the Arctic Ocean — far from areas in which they are used. [22] Like scores of other industrial chemicals, they have also been found in human breast milk, fat and blood.
Fire retardant levels rising rapidly in the environment
Of greatest concern is the exponential rate of fire retardants' increase in the environment. Over the past 20 years, rising levels of PBDEs have been documented by almost every study that looked at trends over time. Earlier this year EWG compared fish caught in San Francisco Bay in 2002 to those caught in 1997 and found PBDE levels in two key indicator species (striped bass and halibut) to be doubling every 2.8 and 3.9 years, respectively. [23] Levels of PBDEs were 100 times higher in San Francisco Bay harbor seals in 1998 than they were 10 years earlier. [24] Similar findings are reported for fish in the Columbia River in Washington State and in the Great Lakes. [25, 26] Around the world researchers have documented similar dramatic increases in wildlife and humans: ringed seals from the Canadian arctic, beluga whales near Baffin Island, blood in the U.S. and Norway, and breast milk in Canada and Sweden. [18, 27, 28, 29, 30]
Research shows that the PBDEs containing 3, 4, and 5 bromines are the most likely of the PBDEs to be absorbed by and build up in living organisms. Penta PBDE is almost exclusively used in flexible polyurethane foam for home and office furniture, carpet padding, and mattresses. About 7.5% of the more than 2.1 billion pounds of flexible polyurethane foam produced in the U.S. each year contains Penta. [31] Fire retardants must be added to foam furniture sold in California, to meet the world's most stringent standards for fire retardancy. [32] Other manufacturers use fire retardants to avoid liability for fire-related injuries. However, the U.S. Consumer Products Safety Commission has said that the same level of fire safety the California furniture standards were designed to achieve can be attained without adding chemical retardants to foam. [33]
PBDEs are thought to enter the human body through exposures to contaminated food, house dust and air. Food is thought to be the major PBDE exposure route for Europeans. The highest levels of PBDEs in foods are typically found in fish. [34, 35, 20] Lower levels are have been documented in chicken [36], other meat and animal fats. [35, 20, 11, 37] Food consumption might not be the dominant source of PBDEs among people in the U.S. and Canada, where the levels of PBDEs found in the human body, household dust, sewage sludge, wildlife and the environment are at least 10 times higher than in other industrialized nations. [38, 39, 40, 41, 42, 43] These findings suggest that inhalation and ingestion of PBDEs in the ambient environment may be significant routes of exposure in North America. [44]
Study findings
Between November 2002 and June 2003, EWG recruited 20 healthy, pregnant women from across the country, all of whom were expecting their first child, to participate in a study of fire retardants in breast milk. Participants collected a breast milk sample within several months of the birth of their child and completed an exposure assessment questionnaire that provided information about their lifestyle and home and work environments. Samples were analyzed by a certified laboratory.
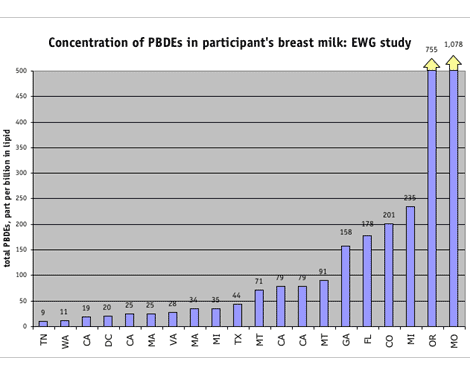
The lab found PBDEs in every breast milk sample tested — 35 different PBDEs in all. More significantly, our tests found levels higher than those reported previously for U.S. women, including two of the highest levels of PBDEs ever reported in human beings in the world. Levels ranged from 9.5 to 1,078 parts per billion (ppb) in milk fat (lipid), with an average level of 159 ppb, and a median value of 58 ppb. Six of 20 participants had PBDE levels above 100 ppb, with two participants exceeding 700 ppb. The highest PBDE level previously reported in the United States was 580 ppb in maternal blood lipid for a woman in Central Indiana. [45]
The most commonly found chemical is known as PBDE-47, which accounted for about half of the total PBDE levels in each participant. It and six other PBDEs accounted for more than 90 percent of each participant's total. Each of these PBDE congeners has three to six bromines and is found in the commercial Penta mixture used predominantly in foam products. There is also evidence suggesting that Deca PBDEs used predominantly in plastics and electronics— which do not readily accumulate in people — are breaking down in the environment to form the congeners that are found in humans.
As observed in other studies of fire retardants, contaminant levels varied widely among participants. [24, 45, 46, 47, 50] All of the participants had higher levels than those commonly detected in European women, and were within the range of body burdens associated with permanent neurological impairments to laboratory animals. Unlike most previous studies of PBDE exposure, we had information about our participants' personal habits, home and work environments. None of the participants reported unusual exposure to PBDE-laden foam or plastics. The variability in PBDE levels we observed in study participants was not easily explained by their diet, occupation, age, body mass or the amount of time they had breastfed their infants.
Of the two most highly-exposed participants, one works in computer-related research and one is a Registered Nurse. One of these participants had replaced the cushions in her sofa and loveseat with cushions purchased at her local variety store. She trimmed the foam cushions for the sofa while inside her home. However, another participant with much lower levels of PBDEs had also recently reupholstered and replaced the foam for a set of dining room chairs, without showing similarly high PBDE levels.
Fish consumption is thought to be a major exposure route for people in Sweden and Finland. [34, 37] One study found that frequent fish-eaters had 5 times higher PBDE levels than non-fish eaters (2.2 vs. 0.4 ppb in lipid.) [34] However, the high PBDE levels in the U.S. might be due to other factors. Seventeen of 20 study participants reported that they typically eat fish at least once per week, and 13 had at least one serving in the seven days before they collected the breast milk sample.
There have been few attempts to characterize fire retardant exposures of the U.S. population. The handful of studies conducted show that PBDE concentrations found in U.S. women and children are the highest reported in the world — and increasing rapidly. Prior to mid-2003 the only information about PBDE levels in U.S. breast milk came from two pooled breast milk samples, collected from an unknown number of women in New York State, Austin, Texas and Denver, Colorado in 1997 and 2000. [48, 49] The samples indicated alarming exposure to PBDEs for U.S. women relative to Europeans and Canadians, but two years passed before a more detailed investigation into PBDE exposures for the U.S. population.
This year, Dr. Arnold Schecter of the University of Texas reported PBDE levels ranging from 6 to 419 ppb lipid for 47 nursing women in Austin and Dallas, the first individual findings for breast milk in the U.S. [47] The study found an average level of 74 ppb and a median (midpoint) value of 34 ppb, 10 to 100 times higher than levels in European women. Total PBDE levels did not vary depending on the mother's age, number of previous births or length of time nursing her infant.
EWG's study found PBDE levels at least 56 percent higher than the Texas and Indiana studies. The average and median (midpoint) values for the most prominent PBDE (PBDE-47) are almost double the levels reported in California women in the late 1990s. [50] This might be in part because we included only first time mothers, who are known to have higher concentrations of many persistent contaminants in their breast milk relative to women who have previously breastfed.
The mean level of PBDEs in our study of women's breast milk was 159 ppb, more than 50 times higher than that reported in a recent Swedish study. [51] In fact, the lowest level of PBDEs in the women we tested (9.5 ppb) was higher than the highest levels measured in Sweden (7.7 ppb), [51] where PBDEs have been voluntarily phased out and will be banned completely next year. [52]
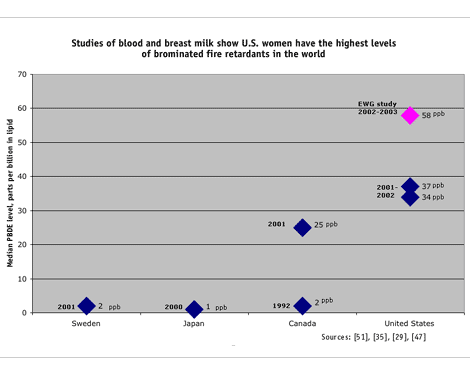
Women in other European countries and in Japan appear to have PBDE levels akin to those in Swedish women. Mothers' milk collected in Finland in the mid-1990s had no more than 6 ppb [53]; in that same period German women had a maximum of 11 ppb [54]; and Japanese women studied in 2000 had no more than 1.5 ppb PBDEs. [35]
Asian and European women's exposures to PBDEs are likely much lower than Americans' because the U.S. and Canada use about 95 percent of the form of PBDEs used in foam furniture, which are known to accumulate most widely in people and in the environment. PBDE levels as high as 590 ppb have been measured in the breast milk of Canadian women, though average levels in a recent study were about 40 ppb — still a fraction of those we detected in U.S. women. [29]
PBDE levels rising rapidly in people
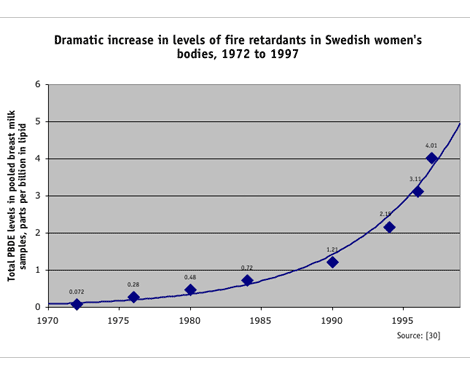
Swedish researchers first reported an exponential increase in PBDE levels in 1999. After detecting a new contaminant in a breast milk sample, researchers examined archived breast milk samples collected over a 25-year span and found an amazing 60-fold increase in the concentrations of PBDEs in breast milk between 1972 and 1997 — equivalent to a doubling every five years. They noted that the increase was startling, given that levels of many persistent chemicals (including PCBs) declined sharply in the same period. [30, 55] The Swedish findings garnered worldwide attention, revealing a global pollution problem, spurring additional studies and a phase-out and eventual ban of most PBDEs in the European Union, beginning in 2004. [52]
Inspired by the work of Swedish researchers, Canadian experts later discovered that PBDE levels in the breast milk of women in Vancouver, B.C. were 15 times higher in 2002 than they had been just 10 years earlier. Not only were PBDE levels rising more quickly in Vancouver than in Sweden, doubling in 2.6 years as opposed to five years, but also average PBDE levels were at least 10 times higher in Canadian women than their Swedish counterparts. [29]
Recent evidence of a time-related increase in PBDE levels in the United States comes from the San Francisco Bay Area. It indicates that PBDE levels in Bay Area women's blood and breast tissue have more than tripled since PBDEs were first introduced in commercial products about 30 years ago. [50] Also, a recent study of archived blood bank samples from the Southeast and Washington state shows a steady rise in PBDE levels in adult blood serum since 1985, and a steadily decline in PCB and PBB levels over the same period. [28]
Exposures to fetuses and babies
Measuring PBDE levels in breast milk is a useful and accurate way to investigate exposures for the developing fetus and newborn baby. PBDE levels are highly similar when measured in a woman's blood serum or in her breast milk or in her infant's umbilical cord blood. [45, 51, 53] Our findings of high levels of PBDEs in breast milk, and recent studies of PBDE levels in infant cord blood, demonstrate that developing babies' exposure to PBDEs is ubiquitous in the United States.
For many years scientists considered the developing fetus to be shielded by the placenta from many chemicals. However there is a growing awareness that virtually any substance present in the mother's body is transported to some extent into the womb. [56] New evidence suggests that most chemicals that accumulate in a mother's body fat can cross through the placenta and be incorporated into the developing infant's body at high levels. Studies of PBDE's cousin, PCBs, indicate that the developing fetus is particularly sensitive to toxic insult. [4] In-utero exposures are dangerous because they occur during a period of dramatic mobilization of maternal fat stores to nurture the rapidly developing fetus especially during the third trimester of pregnancy a time where there is rapid development of the brain and nervous system.
This year, researchers in Central Indiana studied 12 infant-mother pairs to see whether PBDEs were reaching the bodies of developing babies. They found ample evidence that exposures were occurring before birth. Almost identical contaminant levels were measured in maternal and fetal blood. [20] Another study found a similarly strong concordance between levels of PBDE in maternal and cord blood and breast milk samples. The researchers also note that the smaller, more accumulative, and most toxic PBDEs, those with fewer bromines, moved from maternal blood to cord blood most easily. [51]
The high levels of PBDEs in the maternal body and ease of transfer to the developing fetus result in highly exposed newborns. The Indiana-born babies begin their first day of life with PBDE levels ranging from 14 to 460 ppb PBDEs, some 30 to 100 times higher than Swedish newborns [51], and 35 times higher than blood samples collected from adults in Indiana just 15 years ago. [57] Data from these studies show that US babies are born with PBDE levels much higher than European babies.
Exposures through breastfeeding
One of the most meticulous studies to explore adverse health effects resulting from breastfeeding indicates that infant exposure to PCBs via breast milk is much less of a threat than in-utero exposures. This study tracked children over eleven years born to Michigan mothers who ate PCB-contaminated fish during pregnancy. The researchers reported that babies who were breastfed at least six weeks did better than non-breastfed babies on tests designed to measure subtle neurological differences in young children. [4]
The researchers concluded, "Virtually no adverse effects were found in relation to postnatal exposure to PCBs from breast-feeding, indicating that the fetus is particularly vulnerable to this exposure." [4] This may be because the newborn baby has better mechanisms to detoxify chemicals; because the newborn is not as vulnerable to the impacts of PCBs; or because beneficial compounds in breast milk like omega-3 fatty acids protect the developing brain or repair subtle deficits incurred during the prenatal period. [4, 58]
Despite the numerous benefits associated with breastfeeding the detection of PBDEs in breast milk signals dangerous exposures to the developing fetus and baby. A recent study of human serum in Norway found that infants had higher levels of PBDEs and other brominated fire retardants than any other group. [59] Average PBDE levels for infants aged zero to four years were 2.8 times higher than all other age groups. In-utero transfer and breastfeeding are suspected as major sources of PBDE levels in infant serum. Dust is another possible source, because infants ingest more dust than older children or adults, and these fire retardants are detected in high levels in household dust. [39] The evidence that young children are especially exposed and uniquely vulnerable to these toxic chemicals heightens the need for action to limit exposures.
Health risks of PBDEs
A growing body of research in laboratory animals has linked PBDE exposure to an array of adverse health effects including thyroid hormone disruption, permanent learning and memory impairment, behavioral changes, hearing deficits, delayed puberty onset, decreased sperm count, fetal malformations and, possibly, cancer. Research in animals shows that exposure to brominated fire retardants in-utero or during infancy leads to more significant harm than exposure during adulthood, and at much lower levels. And some of these studies have found toxic effects at levels lower than are now detected in American women. Many questions remain, but new evidence raises concerns that low levels of PBDE exposure pose a significant health risk to developing animals, and may pose a health risk to fetuses, infants and children at levels currently detected in American women.
The indication that PBDEs can cause subtle neurological deficits in developing animals echoes what researchers have learned over the past 20 years about the structurally similar, but much better studied, PCBs. Used primarily as electrical insulators, PCBs were found to be rapidly building up in people and animals before they were banned in 1977. Although levels are now declining, PCBs persist in the environment and cause a number of well-documented health problems. Recent studies have shown that PBDEs can act in concert with PCBs and other chemicals through similar mechanisms to increase their effects. [60, 61, 62]
Many of the known health effects of PBDEs are thought to stem from their ability to disrupt the body's thyroid hormone balance, by depressing levels of the T3 and T4 hormones, which are important to normal metabolism. In adults, hypothyroidism can cause fatigue, depression, anxiety, unexplained weight gain, hair loss and low libido. This can lead to more serious problems if left untreated, but the consequences of depressed thyroid hormone levels on developing fetuses and infants can be devastating. [63] One study, for instance, found that women whose levels of T4 measured in the lowest 10 percent of the population during the first trimester of pregnancy were more than 2.5 times as likely to have a child with an IQ of less than 85 (in the lowest 20 percent of the range of IQs) and five times as likely to have a child with an IQ of less than 70, meeting the diagnosis of "mild retardation." [64] An IQ less than 85 can be associated with serious consequences. Two-thirds of children who drop out of high school have IQs below 85.
Even short-term exposures to commercial PBDE mixtures or individual congeners can alter thyroid hormone levels in animals, and the effects are more profound in fetuses and young animals than in adults. [65, 66, 67, 68, 69, 70] These results aren't surprising, but are ominous as data in humans indicate that pregnancy itself stresses the thyroid, and developing fetuses and infants do not have the thyroid hormone reserves adults do to help buffer insults to the system. [71]
Most studies on thyroid hormone disruption by PBDEs have been short-term, with exposures of 14 days or less. The real question is how low doses over the long term affect the body's thyroid hormone balance. The answer is important, because the entire U.S. population is exposed daily to low levels of PBDEs, and studies of other thyroid hormone disrupters have found that long-term exposures can cause more serious harm at lower levels of exposure. [72] Although no direct link could be made, one study found higher rates of hypothyroidism among workers exposed to brominated fire retardants on the job. [73]
Because the developing brain is known to be extremely sensitive to neurotoxicants, researchers have begun to examine whether short-term exposures to PBDEs at critical times could have long-term effects. The results are troubling: small doses administered to fetal or newborn mice and rats caused deficits in learning, memory and hearing, changes in behavior, and delays in sensory-motor development. Many of these effects were found to worsen with age, and the effects were seen with the higher-weight PBDEs (the usually less harmful Deca) as well as the more readily absorbed lower-weight congeners.[7, 74, 75]
Harm at one dose?
Experiments have shown that just one dose of PBDEs at a critical point in brain development can cause lasting harm. [7, 74, 75] In two different studies, a small dose — as little as 0.8 milligrams per kilogram of body weight per day (mg/kg-day) — given to 10-day-old mice caused "deranged spontaneous behavior," significant deficits in learning and memory and reduced ability to adapt to new environments, with these problems often becoming more pronounced with age. [7, 75] This research also demonstrated the heightened sensitivity of the brain at certain critical phases of development. While earlier exposures caused "significantly impaired spontaneous motor behavior" and "persistent neurotoxic effects," no effects were seen in mice that were exposed later in development, despite having similar levels of PBDEs (or their metabolites) in the brain. [7]
Other animal studies have shown that early-life exposures to PBDEs, often at relatively low levels, can lead to delays in sensory-motor development, hearing deficits, as well as changes in activity levels and fear responses. [74, 76, 77] At this point, scientists do not understand exactly how PBDEs affect neurological development. But there is evidence that PBDEs and/or their metabolites are in fact acting through several different mechanisms, including mimicking thyroid hormones, increasing their rate of clearance in the body and interfering with intracellular communication. [78]
In addition to their effects on thyroid hormones and neurological development, PBDEs have been linked to a gamut of other health impacts in laboratory animals, from subtle to dramatic. For example, several new studies found that early-life exposure to PBDEs has significant reproductive effects including delaying the onset of puberty in male and female rats and decreasing the weight of male rat reproductive organs and sperm count. [79, 80, 81, 82] In studies of pregnant animals, PBDE exposure was associated with retarded weight gain, enlarged livers and raised serum cholesterol. [83, 84] In-utero exposures have also been associated with serious harm to the fetus, including limb and ureter malformation, enlarged hearts, bent ribs, fused stemebrae, delayed bone hardening, and lower weight gain. [83, 84, 85, 86] The malformations of the fetus were consistently seen at levels much lower than doses harmful to the mouse mothers — the lowest being 2 and 5 mg/kg-day, respectively. But more subtle reproductive effects, such as decreased sperm count and changes in the sub-cellular structure of the ovaries, were seen at incredibly low doses — just 0.06 mg/kg-day. [82]
The few studies that have looked at changes in organ structure have found that semi-chronic PBDE exposure can cause thyroid hyperplasia (overgrowth of thyroid tissue) and enlarged livers at relatively low doses (10 mg/kg-day) and other adverse effects such as abnormal cell functioning, localized cell death and deformation in the kidney, changes in the liver's cellular structure, decreased hemoglobin and red blood cell counts at higher doses. [83, 85, 87, 88] Only one commercial PBDE mixture has been tested for its ability to cause cancer, in a single study more than 15 years ago. High doses of Deca given to rats and mice caused liver, thyroid and pancreas tumors. [88] Deca-BDE is the least easily absorbed and the most rapidly eliminated of the PBDEs, and recent research indicates that other congeners can cause genetic recombination in cells, which raises concern for carcinogenicity. [89] As a result, scientists believe that the congeners with fewer bromines are likely to be more carcinogenic than deca-BDE and have urged that such tests be conducted. [78]
PBDEs cause adverse effects in rodents at low doses.
|
PBDE congener |
Test animal |
PBDE concentration in fat tissue |
PBDE dose (mg/kg-day) |
Toxic effect |
Source |
|
PBDE-99 |
Mice |
12 ppb in brain lipid* |
0.8 (single dose) |
Effects on learning and memory, spontaneous motor behavior and habitutation capability that worsened with age |
[75, 7] |
|
Commercial Penta Mixture (DE-71) |
Mice |
Not measured |
0.8 (single dose) |
Significant decrease in thyroid hormone (T4) levels |
[65] |
|
PBDE-99 |
Rats |
Not measured |
0.06 (single dose) |
Decreased sperm count |
[82] |
|
PBDE-99 |
Rats |
Not measured |
0.06 (single dose) |
Changes in the subcellular structure of ovaries |
[113] |
Levels of fire retardants in breast milk detected in some participants are higher than levels in mouse brain lipid levels that have been linked to adverse health effects. The relationship between contaminat levels in the lipid of breast milk and brain tissue is not known.
* Assuming that a rodent brain is 30% lipid [8]
PBDE body burdens nearing threshold for harmful effects
A growing body of animal research shows a very low threshold for PBDEs to cause permanent impacts to the nervous system. One of the lowest harmful doses of PBDEs was found in a 2002 study of newborn mice which showed neurodevelopmental damage at concentrations of just 4 ppb in brain tissue or about 12 ppb in brain fat. This study exposed lab animals to a single dose of one type of PBDE, called PBDE-99. Thirty percent of the participants in our study and almost 20 percent of women in the Texas study had more than 12 ppb of PBDE-99 in the fat of their breast milk, but no studies have investigated the relationship between contaminant levels in lipid of brain tissue and lipids from breast milk or other body tissues, making it difficult to determine if human exposures exceed levels known to permanently damage rodent brains.[7, 8] Scientists are most concerned about the neurological impacts of PBDEs on the fetus and young child. These impacts are inherently difficult to detect in rodent studies, which can't measure subtle impacts to learning, memory and behavior. The fact that we are seeing these effects in lab animals heightens concerns for human health. [90]
A scientist at the California Environmental Protection Agency's Office of Environmental Health Hazard Assessment (OEHHA) combined all the data on PBDE body burdens in tissue, blood and breast milk from women across the country available in April 2003 to project PBDE levels in the entire U.S. population. If the data used in the model are representative of the American population, as many as 15 million Americans would have a body burden more than 400 ppb for all PBDEs. [8] Our study measured much higher contaminant levels than those included in the model, indicating that these calculations might underestimate exposures to the American public.
PBDEs have been shown to impact the same body systems as their notorious cousins, the PCBs. [7, 62] For example, a recent study showed that a low dose of a single PBDE congener given to pregnant rats along with a low dose of a single PCB conger caused neurodevelopmental deficits in offspring that were not seen when the same doses of the congeners were given alone. [60]
While PCB levels are declining, they still harm infant development at levels commonly detected in the American population. Current body burdens of PBDEs in U.S. mothers' breast milk are thought to be slightly lower than levels of PCBs, and PBDEs are thought to be less potent toxicants. However, the rapid increase in PBDE levels and significant number of highly-exposed people, both indicate that PBDEs could soon eclipse PCBs as the predominant persistent toxin in our bodies. If the increase in the fire retardant body burdens continues at its current rate, levels of PBDEs will soon exceed levels of PCBs associated with a six-point deficit in IQ points in children (>1,250 ppb in lipid.) [91]
If fire retardant body burdens are found to be causing adverse neurological effects, either alone or in concert with exposures to other common bioactive chemicals, the potential costs of this damage could be significant. Government agencies considering protection and standards for lead and PCBs have attempted to calculate the societal costs of reduced learning capacity — effects associated with exposure to these chemicals for young children. They attempt to quantify the impact to society in terms of increased special education and reduced lifetime earnings capacity, as measured by subtle deficits in learning and memory measured by IQ and other tests. Their estimates vary widely depending on the impact measured. A one-point loss in IQ over an entire population of newborn children in the United States has been calculated to cost $55 to 65 billion per year. [92] Given that learning, developmental and behavioral disabilities already affect nearly 12 million U.S. children [93], it is prudent to control avoidable sources of contamination that threaten permanent effects on our children's health.
Failure to protect children
Evidence of PBDE contamination in women's bodies and breast milk in the US should come as no a surprise. The evidence against PBDEs was strong enough that bans were proposed in Germany, Sweden and the Netherlands in the mid-1980s and early 1990s. Industrial users of the chemicals agreed to voluntarily phase them out in Germany in 1986, with the manufacturers and users in the other two countries later following suit. In 1993, Germany placed official limits on PBDE use under its Dioxin Ordinance because of the tendency for PBDEs to release dioxins when burned. [94] As concern spread to other countries, the European Union launched a scientific review of the safety of PBDEs, originally with respect to electronics waste. In February 2003, the EU announced a ban on two common PBDE mixtures (Penta and Octa) in all products as of August 2004. [52] The EU is also considering a ban on Deca for use in electronic products by July 2006. Pending the completion of further studies, the EU Chemicals Inspectorate will decide whether to also ban Deca in other non-electronic products as of 2006. [95]
Even before the ban takes effect, the earlier efforts to reduce PBDE use in Europe are paying off. Researchers have found that PBDE levels in Swedish breast milk rose exponentially from 1972 to 1997, but since that year have begun to decline: PBDE levels in Swedish women dropped about 30 percent between 1997 and 2001. [96] These results are encouraging. This shows that if protections are enacted and PBDE use ceases or declines, the human body burden of PBDEs will also decrease after a lag-time of several years or more. However, given the massive amount of PBDEs in U.S. homes, and the much higher levels than in European women, PBDE body burdens in Americans might take longer to decline.
Despite that fact that PBDE concentrations in Americans and their environment are ten to 100 times higher than those found in Europe, the U.S. government has so far done nothing to counter this rapidly escalating problem — nor did it do much to stop it in the first place, when safety testing could have flagged the problem in its infancy. Like almost all industrial chemicals, the health effects of PBDEs went virtually unstudied at the dawn of their use in commercial products. In 1994, EPA determined that the waste stream from the production of Octa and Deca "should not be listed as hazardous." [97] The only other standard governing PBDEs is the requirement that companies that manufacture or use large amounts of Deca report their chemical pollution under the Toxics Release Inventory.
State action a first step
State legislation was introduced in California, Maine and Michigan this year that would ban or restrict the use of several types of these fire retardants. While these bills are a welcome first step, they all fall short of what is needed to prevent further build up of these persistent, bioaccumulative and toxic chemicals.
A bill passed by the Maine legislature (LD 743), and signed into law in May, 2003 requires electronic manufacturers or importers to phase out all brominated fire retardants and several other toxic substances such as lead, mercury and polyvinyl chloride (PVC) by 2006. But producers can apply for exemptions if they can convince officials that there are no feasible alternatives. Of course, the law does nothing about the brominated fire retardants in non-electronic products. Bills that would have banned the use of PBDEs in all products by 2006 were introduced in Michigan during 2003 and 2002 (HB 4406 and HB 5575, respectively), yet have failed to progress in the legislature.
The California standards, signed into law in July 2003, ban two commercial mixtures, Penta and Octa, but exempt the most widely used PBDE product (Deca-discussed below). This is troublesome, as numerous studies have shown that the types of PBDEs in this commercial product can break down into other congeners that are much more bioaccumulative and bioreactive, and which are included in the proposed California legislation. The law also gives PBDE producers and users until 2008 to stop using the chemicals, despite the fact that another 365 million pounds of PBDEs will be put into American couches, easy chairs, cars, planes, buses and other consumer products before the phase-out date in five years. [10] Finally, the law doesn't require manufacturers to label PBDE-containing products, a provision that would have allowed consumers to make more informed decisions, rewarded the companies who have already shifted away from PBDEs, and provided extra incentive to manufacturers and users to speed their conversion to new fire retardants, materials, or design.
What about the other PBDEs?
One of the major debates about regulation of PBDEs centers on the effects of the various congeners in the environment. Scientists have found that PBDEs with five or fewer bromines are almost totally absorbed by the body, slowly eliminated, highly bioaccumulative, and cause health effects at relatively low levels. In contrast, PBDEs with more bromines are less readily absorbed, less bioaccumulative, more quickly eliminated by the body, meaning that they are found at lower levels in living creatures. [32]
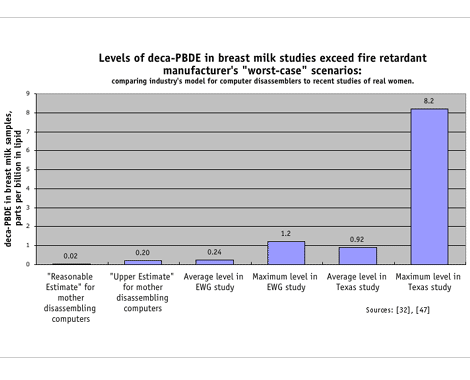
Fire retardant manufacturers have claimed that Deca is "very poorly absorbed," (less than 2 percent of an oral dose is absorbed) and rapidly excreted (with almost complete excretion within 72 hours). [32]
Thus, it would be virtually impossible for Deca to enter women's bodies and even more unlikely for it to pass into breast milk. [32] Despite these assurances, measurable levels of Deca were detected in 16 of 20 young mothers at levels up to 1 ppb, as well as eight of 23 women in Dr. Schecter's recent Texas study — at concentrations as high as 8 ppb. [47] The laboratories used in the studies cannot yet test for octa-BDEs in breast milk or other biological samples.
Fire retardant manufacturers have estimated the concentrations in breast milk of a mother working to disassemble computer monitors. They modeled two scenarios, the "Reasonable Estimate" using "plausible, yet conservative" assumptions and the "Upper Estimate" using "absolute worst-case exposures." [32]
They concluded: "[A] significant health risk is not expected for children under any of the scenarios evaluated, even using extremely conservative assumptions. Therefore, no further, more detailed evaluation of [Deca] is warranted to ensure adequate health protection for young children."
Maybe the industry should take a closer look at the everyday exposures for American mothers before they claim that their product is safe. In fact, in two recent studies of just 67 participants, we found levels of Deca in young mothers who were not occupationally exposed to be 6 to 40 times higher than industry's model for women disassembling computers at work.
Toxic effects of Deca
New research also indicates that Deca may be more toxic than previously thought, exhibiting some of the same effects on newborn rats and mice as PBDEs with fewer bromines. [98] And equally importantly Deca can covert to PBDEs with fewer bromines which are more toxic and more likely to accumulate in human beings. Numerous studies show that when exposed to sunlight, the higher-weight congeners can be converted to the more toxic PBDEs with fewer bromines. [99, 100, 101, 102, 103] This degradation in the environment has been observed for structurally similar chemicals like polybrominated biphenyls (PBBs) and PCBs. In laboratory conditions Deca can degrade to 50 percent of its original mass in as little as 15 minutes, [99] and one study found that after five days of exposure to sunlight just 6 percent of the deca-BDE congener remained undegraded. [104]
The bottom line is that all chemicals in the PBDE family have the potential to cause serious environmental and health problems — some alone, some through their breakdown products, others by interacting with other toxic chemicals, and all by interfering with important life processes in the environment, wildlife and humans. The chemical industry, trying to save a highly profitable product (Deca), is pushing the notion that certain PBDEs are harmless. The available evidence argues the opposite: in the environment, Deca degrades into the very chemicals being banned in Europe and California. To prevent a bad situation from getting worse, all PBDEs should be phased out without delay.
Affordable Replacements
For most uses of brominated fire retardants there are already chemical replacements on the marketplace at equivalent or only slightly higher cost. Aluminum trihydroxide and various phosphorous-based compounds are some of the most common alternatives. But rather than replacing one chemical with another that is unstudied and might also be toxic. A better solution is to redesign products so that chemical fire retardants are not needed to meet fire safety regulations.
The U.S. Consumer Products Safety Commission recently reported that adding fire retardants to foam offered very little additional protection from fire: "CPSC laboratory tests have demonstrated that the properties of actual filling materials have little or no effect on the small open flame ignition resistance of full-scale chairs." [33] Simply using a barrier fabric and less flammable foam can achieve the same level of fire safety. Similarly, increasing the density of polyurethane foam or using materials that are naturally less flammable can eliminate the need for chemical fire retardants. [105]
Another non-toxic tactic to prevent fire is to control sources of fire ignition. One target is cigarettes, which are responsible for the bulk of fatal blazes. New York state blamed cigarettes for 199 deaths between 1997-2001, making smoking materials the most frequent cause of fatal blazes during that period. [106] In response, the state passed legislation to require that cigarettes sold by mid-2004 must be self-extinguishing. This can be easily accomplished by wrapping the tobacco in a heavier paper and removing the citrates added to cigarette wrappers to promote burning. The tobacco industry is expected to fight the legislation in court.
Trade groups and fire retardant producers such as the California Manufacturers and Technology Association, California Retailers Association, the American Electronics Association and Great Lakes Chemical vigorously opposed the California ban bill. However, facing restrictions on PBDE use in the European Union, many U.S. manufacturers moved to find alternatives to PBDEs, even in the absence of national regulation. Computer and electronics companies such as Apple, Dell, IBM, Motorola, Panasonic, Phillips and Sony are already producing some PBDE-free or BFR-free products. [105] Ericsson, Intel, Phillips, Sony and Toshiba recently announced a complete ban of PBDEs in advance of the EU regulation by 2006. IKEA and Ericsson have taken additional steps toward moving away from using any halogens (bromine, chlorine, fluorine or iodine) in their products. [105]
Fire retardant manufacturers are scrambling to find a replacement for Penta used in foam furniture. The Swedish furniture giant IKEA was forced to phase out brominated fire retardants in all its products due to European limits on the chemical's use. They accomplished this by changing product design, using naturally less-flammable materials, and employing alternative fire retardants if needed. Hickory Springs of Conover, N.C., a major polyurethane foam producer, is working with Akzo Nobel, a chemical manufacturer, to test a non-halogenated, phosphorous-based fire retardant. Hickory Springs says it was motivated by requests from companies such as IKEA, Crate & Barrel and Eddie Bauer for PBDE-free furniture. [107]
No safety studies on many toxic chemicals
Several US states have taken important steps to phase-out a handful of brominated fire retardants. This is the first step toward protecting consumer safety, but it offers incomplete protection as long as manufacturers are not required to test the impact of replacement chemicals on human health — before they go into nationwide use. There is very little data on the toxicity of the fire retardants that are currently being developed or are already in use as alternatives to PBDEs. This is largely because of well-documented shortcomings in the nation's toxics laws. The chief regulatory statute for commercial chemicals, the Toxic Substances Control Act (TSCA), is infamous for its failure to lend meaningful authority to the Environmental Protection Agency. [108] The looming PBDE crisis and uncertainty surrounding replacement chemicals provide another disturbing illustration of the failures of a regulatory system that allows persistent, bioaccumulative toxins onto the marketplace before they have been adequately tested for safety. With these fire retardants, we are again reaping the high costs, in terms of health and productivity, of this industry-favored system.
Under the current system, the EPA reviews new chemicals through a process that does not require health and safety test data and that discourages voluntary testing. Companies submit only basic toxicity data for fewer than half of all applications for new chemicals, and the government approves 80 percent of these with no use limits and no requests for tests. Eight of 10 new chemicals win approval in less than three weeks, at an average rate of seven a day. [108]
Perhaps worse than our weak controls on new chemicals is the blind eye we turn on the old ones. When TSCA was enacted in 1976, more than 63,000 chemicals already in use were "grandfathered" — granted blanket approval for continued use in consumer and industrial products with absolutely no requirement for further study. Most brominated fire retardants fell into this loophole and won implicit approval for widespread use in consumer products with no required health and safety testing. In 1998, the EPA and the nonprofit organization Environmental Defense reviewed all of the toxicity and environmental fate studies publicly available and found no information — not a single test — for 43 percent of the 2,600 chemicals produced in the highest volumes in the U.S. [108, 109]
The chemical industry has since agreed to do more tests to assess potential toxicity to children for a select number of the most widely use chemicals under the Voluntary Children's Chemical Exposure Program (VCCEP). The three most widely-used PBDE mixtures were included in the first group of 23 chemicals to be assessed as part of this program, but the usefulness of the VCCEP program is highly limited. Its purpose is to make "health effects, exposure, and risk information" of these chemicals available and provide "the means to understand the potential health risks to children." [110] But because the program is voluntary, chemical manufacturers are unlikely to hand over any information that might be damning for their chemical products, nor do they have much incentive to fill any significant scientific data gaps that are identified in the process.
There is no question that fire safety is important and that making products fire-resistant can save lives. Chemical fire retardants have become ubiquitous over the last few decades, but a wide variety of fire safety strategies exist. Using less-flammable materials or changing the product design so that it is inherently more fire resistant, are chemical-free solutions. Using less toxic chemicals as fire retardants is another option.
We do not have to expose our children to toxins to protect them from fire.
Need for biomonitoring
The current system for biomonitoring in the U.S. is highly inadequate for identifying and tracking the multitude of chemicals Americans are accumulating and carrying in our bodies. The Centers for Disease Control and Prevention (CDC) has conducted two biomonitoring studies that examined up to 116 environmental chemicals in the blood and urine of 2,500 people. [111] But this is just a tiny subset of the thousands of chemicals we are exposed to every day, and will always have a lag-time of several years or more when investigating news of emerging contaminants of concern such as PBDEs or any other brominated fire retardants. Furthermore, the CDC study measured only four chemicals in children younger than six years old and did not look at any contaminants in breast milk, both of which are important for estimating chemical exposures to these most sensitive subpopulations.
In an age where chemical industries are releasing millions of tons of chemicals to the environment and chemical manufacturers gain permission to put more than 2,000 new chemicals into the biosphere each year, we desperately need a better biomonitoring system. Such a system would serve as an early warning system for chemicals that are building up in our bodies, that can track trends in chemicals levels over time, and most importantly, trigger prompt regulatory action when necessary.
What you can do
EWG's tests of mothers' milk are the latest evidence that Americans are being exposed to potentially harmful levels of toxic fire retardants. The bad news is that efforts by both government and private industry in the U.S. are lagging behind Europe, which has already phased out some fire retardants and is studying the toxic effects of others. The good news is that European studies show that levels of fire retardants in the human body begin to decline relatively quickly if exposure is reduced. That means that prompt action by government agencies and the companies that make or use fire retardants can make a difference. To some extent, personal actions can also reduce your exposure.
What should government do?
- The U.S. EPA should phase out all PBDEs and other toxic fire retardants as quickly as possible. California has already moved to ban some PBDEs in 2008, and Massachusetts is considering a similar law. In the interim, all products containing PBDEs should be labeled so that consumers have the option of choosing products without them.
- EPA must screen all new and existing chemicals for their health effects. In particular, potential replacement fire retardants must be adequately tested to ensure that they are not persistent, bioaccumulative or toxic. Testing must include the outcomes most relevant to children's health. Changes in product design that decrease the need for chemical fire retardants should be encouraged over simply switching to a different, less studied chemical.
- The U.S. Centers for Disease Control and Prevention should expand the fledgling national biomonitoring program to include a greater number of chemicals and people. The study provides critical data in identifying chemicals that are accumulating in our bodies and in the environment; tracking trends in exposure; providing data needed to more fully understand human health risks; and helping EPA and other agencies effectively transition businesses to safer, less persistent chemicals than those in current common use.
- Congress should increase funding for urgently needed additional research on toxic fire retardants, including their health effects, how they get into the human body, and current levels of accumulation in people, animals and the environment.
What should private industry do?
In the absence of government regulation, U.S. manufacturers and users of chemical fire retardants should voluntarily comply with the European ban. Chemical companies should work to minimize the toxicity of existing fire retardants and thoroughly test replacement chemicals for safety. Companies who use fire retardants in their products should follow the lead of some computer makers, who are redesigning their products so that fire retardants are not needed. Retailers should follow the example of IKEA and some other companies in demanding that their suppliers avoid the use of chemical fire retardants.
What should parents and other concerned consumers do?
Our homes and offices are filled with brominated fire retardants in products including foam-padded furniture, computer and television screens, and the padding underneath our carpets. Our study found that exposure to brominated fire retardants is unavoidable. We detected them in the body of every participant, regardless of their occupation, diet, or lifestyle.
Even if these toxic fire retardants were phased out immediately, our exposures to them would continue through the foods we eat or from the products in our households. In the absence of government safeguards to remove persistent toxins from household products, or label products containing the most toxic forms of fire retardants, parents should consider the following options:
- Avoid degraded or crumbing foam that might contain fire retardants. Replace or cover couches, stuffed chairs, automobile seats that have exposed foam. Do reupholster padded furniture in homes where children or pregnant women live.
- Be careful when removing and replacing the foam padding beneath your carpet. Remove old carpet padding from your home and clean up well when finished.
- Buy products with natural fibers (cotton and wool) which are naturally fire resistant.
Many other persistent pollutants, some banned for decades, still contaminate the environment and end up in the food we eat, the water we drink, and the air we breathe. Recently, EWG has reported on the presence of toxic chemicals in a wide range of consumer products including foam-padded furniture, food wrappers and winter-time lettuce. In the case of toxic fire retardants, chemical companies have fought proposals that they label their products to give consumers information about the chemicals in consumer products.
Yet exposures to many persistent pollutants can be reduced through a varied diet that contains fewer meat and high fat dairy products. Other chemical exposures, like toxic substances in household cleaners, can be avoided altogether. It is especially important for children, pregnant or breastfeeing mothers or women considering pregnancy to avoid chemical exposures. Some simple tips for reducing exposures to, or impacts of, industrial chemicals are:
- Breastfeed your child! Breastfeeding offers significant health benefits to both mother and infant. In addition, breast milk contains beneficial compounds such as Omega-3 fatty acids that are not found in infant formula and support optimal infant development, particularly for body systems most affected by PCBs, lead, and other toxic chemicals.
- Eat fewer processed foods, which often contain chemical additives.
- Eat organic produce. It's free of pesticides and preservative chemicals.
- Don't microwave food in plastic containers. Use glass or ceramics.
- Run your tap water through a home filter before drinking. Filters can reduce levels of common tap water pollutants.
- Eat fewer meat and high fat dairy products, which contain higher levels of some pollutants.
- Reduce the number of cosmetics and other personal care products you use, which can contain harmful chemicals and can be sold with no safety testing.
- Avoid artificial fragrances.
- Don't use stain repellants on clothing, bedding or upholstery.
- Reduce the number of household cleaners you use. Try soap and water first.
- Avoid using gasoline-powered yard tools — use manual or electric tools instead.
- Avoid breathing gasoline fumes when you're filling your car.
- Eat seafood known to be low in PCB and mercury contamination, including wild Alaska salmon and canned salmon. Avoid canned tuna — it contains mercury.
Breast milk is still best
Evidence of the accumulation of chemicals in women’s bodies and breast milk may prompt mothers to question the safety of their breast milk as a food for their baby. However, the evidence is clear: Women should breastfeed their children and continue to do so for as long as possible. Breastfeeding provides significant health benefits to both mother and child. In fact, careful study of babies’ toxic exposures indicates that it might be even more important for mothers who are concerned about their exposure to toxic chemicals to breastfeed their babies.
Physicians investigating the hazards of chemical exposure via breast milk and the benefits of breastfeeding consistently support breastfeeding as the healthiest way to feed a child. [1, 2, 3] For both baby and mother, breastfeeding has many well-documented health benefits:
- Breastfed babies have lower rates of some of the most serious chronic diseases: asthma [4], diabetes, [5] and some childhood cancers. [6, 7]
- Breastfeeding reduces the risk and severity of communicable diseases: pneumonia, diarrhea, and ear infections. [2, 8]
- Women who breastfeed have lower levels of ovarian and breast cancer, and breastfed daughters also have lower rates of breast cancer when they grow up.
- Breastfeeding may even reverse some of the damage caused by chemical exposures in the womb.
Even with chemicals, breastfeeding is best
Breast milk is made up of fat from a mother’s body. Therefore it contains the same chemicals that accumulate in her body fat, such as PCBs and PBDEs. The developing baby is exposed to chemicals from their mother’s body from pregnancy until it is weaned. Even though breastfed infants are exposed to higher levels of chemicals over their first few years of life, they have lower levels of childhood cancers, breast cancer and other illnesses believed to be linked to chemical exposure.
Several long-term studies have followed groups of babies exposed to PCBs in-utero and found that the breastfed babies appear to be less impacted by the chemical exposures than their bottle-fed counterparts. [9, 10] One study of Michigan babies found significant improvements in babies’ breastfed for at least 6 weeks. The researchers concluded that PCB exposures in the womb were responsible for the neurological impacts, and that breastfed infants showed fewer effects of PCB exposure. [9]
Despite evidence that breastfeeding can protect against subtle health effects caused by in-utero chemical exposures, we should still be cautious with our children’s exposure to chemicals. Studies documenting extremely high levels of PCBs in Inuit children eating a subsistence diet have found that breastfed infants get as many ear infections as formula-fed babies, indicating that the PCBs may reduce some of the protective benefits of breastfeeding. [11] Another concern is that chemicals mimicking estrogen in a women’s body seem to make it difficult for her to produce breast milk, resulting in earlier weaning of children. [12, 13]
Concerns with infant formulas
Parents concerned about chemical contaminants should also consider the concerns associated with feeding infants formula rather than breast milk:
- Mixing powdered formula with drinking water can expose children to chemical or bacterial contaminants. EWG has reported that infants under four months of age get more than seven times the dose of chemicals in tap water than an adult would get, relative to their body weight. Widespread contaminants of potential concern in drinking water include chlorine byproducts, pesticides, solvents, nitrates, lead, and arsenic. [14] Localized contaminants can also result in risk. As a result of widespread atrazine contamination in the Midwest, EWG estimated that 146,000 infants are born each year in regions with measurable atrazine in their drinking water supply, which provide a bottle-fed infant by age one with 25 percent of his or her lifetime allowable dose of atrazine. [15]
- Breast milk is estimated to contain 160 fatty acids that are not included in baby formula. [16] These fatty acids have been linked to optimal brain development and better vision in breastfed babies. Two in particular have recently begun to be added to formulas-albeit at extra cost. “Superbaby formulas” supplemented with two omega-3 fatty acids, known as docosahexaenoic acid (DHA) and arachidonic acid (A.A.) are now available. [17]
- There are unresolved concerns about the safety of manganese found in baby formula. Manganese is a neurotoxic chemical found in much higher concentrations in infant formula than in mother’s milk. In fact, soy-based formulas contain about 80 times more manganese than breast milk, and formulas made with animal protein about 30 times more. Studies conducted as early as the 1970s and 80s show an association between various learning or behavior problems and elevated manganese levels. [18] Infants are not able to absorb and excrete excess manganese during their first year of life, a period of rapid development. Developmental deficits have been reported in primates fed 50 to 100 ounces of Isomil per day. [19]
Protecting children’s health
In all but the most extreme circumstances, then, breast milk remains the best food for babies. [3] Yet we cannot ignore the increasing burdens of persistent contaminants in the bodies of mothers and children. Without knowing what chemicals are found in our bodies and our homes, or having a reasonable idea that these chemicals are safe, we have no way to protect our infants or children from exposure to toxic chemicals while they are in the womb or subsisting on mother’s milk. Chemicals like PBDEs and their replacements must be thoroughly tested for their safety, before they enter our homes, our environment, and our bodies.
References:
[1] Greater Boston Physicians for Social Responsibility. 1999. What about breastfeeding? http://psr.igc.org/breastfeeding.htm
[2] Greater Boston Physicians for Social Responsibility. 2002. Out of Harm’s Way: Preventing Toxic Threats to Child Development. Why breast-feeding is still best for baby. http://www.psr.igc.org/breasthealth5.30.01_links.pdf
[3] American Academy of Pediatrics (AAP). 1999. Polychlorinated biphenyls, dibenzofurans, and dibenzodioxins. In: Etzel RA, Balk SJ, editors. Handbook of Pediatric Environmental Health. p. 215-22.
[4] Oddy WH, Holt PG, Sly PD et al. 1999. Association between breast feeding and asthma in 6 year old children: findings of a prospective birth cohort study. British Medical Journal 319(7213):815-819.
[5] Mayer EJ, Hamman RF, Gay EC et al. 1998. Reduced risk of IDDM among breast-fed children. Diabetes. (37)1625-1632
[6] Davis MK. 1998a. Review of the evidence for an association between infant feeding and childhood cancer. International Journal of Cancer, Suppl (11)29-33.
[7] Davis MK, Savitz DA, Graubard BI. 1998b. Infant feeding and childhood cancer. Lancet. (2)365-368.
[8] American Academy of Pediatrics (AAP) 1997. Breastfeeding and the Use of Human Milk. Pediatrics Vol. 100(6):1035-39
[9] Jacobson, J.L.; Jacobson, S.W. 2002. Association of prenatal exposure to an environmental contaminant with intellectual function in childhood. Journ. Clin. Tox. 40(4): 467-75.
[10] Dekoning, E.P. and W. Karmaus. 2000. PCB Exposure in-utero and Via Breast Milk, A Review. Journal of Exposure Analysis and Environmental Epidemiology 10:285-293.
[11] Dewailly E, Ayotta P, Bruneau S, et al. 2000. Susceptibility to infections and immune status in Inuit infants exposed to organochlorines. Environmental Health Perspectives 108(3):205-211.
[12] Gladen BC, Rogan WJ. 1996. DDE and shortened duration of lactation in a northern Mexican town. Am J Public Health. Apr;85(4):504-8.
[13] Rogan WJ, Gladen BC, McKinney JD, Carreras N, Hardy P, Thullen J, Tingelstad J, Tully M. 1987. Polychlorinated biphenyls (PCBs) and dichlorodiphenyl dichloroethene (DDE) in human milk: effects on growth, morbidity, and duration of lactation. American Journal of Public Health. (77)10:1294-7.
[14] Natural Resources Defense Council (NRDC). 2001. Healthy Milk, Healthy Baby: Chemical Pollution and Mother's Milk. http://www.nrdc.org/breastmilk
[15] Environmental Working Group. 1999. Into the Mouths of Babes: Bottle-fed Infants at Risk from Atrazine in Tap Water. https://www.ewg.org
[16] Kaufman, M. 1999. FDA revising regulations to improve baby formula. Washington Post September 14, 1999.
[17] Retsinas, G. New York Times. 2003. The Marketing of a Superbaby Formula. June 1, 2003.
[18] Collipp PJ. Manganese in infant formula and learning disability. Ann Nutr Metab 27:488-494. 1983.
[19] Van Scoy, H. Soy-based formulas may be linked to ADHD: elevated levels of manganese the suspected culprit. Health Scout News Reporter. October 8, 2002. http://www.hon.ch/News/HSN/509534.html
PBDE Health Effects: Independent Studies
Dozens of independent studies have linked low levels of bromated fire retardants with a variety of adverse health effects with more being published in the near future. Listed below is a small subset of these studies with a summary of their key findings and links to their abstracts where available.
• Eriksson P, Jakobsson E, Fredriksson A. Brominated flame retardants: a novel class of developmental neurotoxicants in our environment? Environ Health Perspect. 2001 Sep;109(9):903-8.
Key finding: A single dose of PBDEs given to mice in early development caused effects on learning and memory, spontaneous motor behavior and habitutation capability that worsened with age.
• Branchi I, Alleva E, Costa LG. Effects of perinatal exposure to a polybrominated diphenyl ether (PBDE 99) on mouse neurobehavioural development. Neurotoxicology. 2002 Sep;23(3):375-84.
Key finding: A low dose of PBDEs given to mice in early development led to changes in behavior.
• Eriksson P, Viberg H, Jakobsson E, Orn U, Fredriksson A. A brominated flame retardant, 2,2',4,4',5-pentabromodiphenyl ether: uptake, retention, and induction of neurobehavioral alterations in mice during a critical phase of neonatal brain development. Toxicol Sci. 2002 May;67(1):98-103.
Key finding: A single dose of PBDEs given to mice in early development significantly impaired spontaneous motor behavior.
• Zhou T, Taylor MM, DeVito MJ, Crofton KM. Developmental exposure to brominated diphenyl ethers results in thyroid hormone disruption. Toxicol Sci. 2002 Mar;66(1):105-16.
Key finding: PBDEs are an endocrince disruptor during development.
• Kuriyama, S. and Chahoud, I. 2003. Maternal exposure to low dose 2,2'4,4',5 pentabromo diphenyl ether (PBDE 99) impairs male reproductive performance in adult male offspring. Organohologen Compounds. (61):92- 95.
Key finding: Single very low dose of PBDEs given to rats in early development caused decreased sperm count in adult offspring.
Abstract not available on-line. EWG can provide copy upon request.
Appendix 1: Methods & sample analysis
Sample collection
Between November 2002 and June 2003, EWG recruited 20 healthy, breastfeeding mothers from 14 states to participate in a breast milk study. Participants collected a breast milk sample into chemically clean, 4 oz study jars, between 7 and 100 days postpartum. Ten participants hand expressed the breast milk sample and ten participants used sterilized, commercially available breast pumps. Study jars were frozen, packed with ice and sent overnight delivery to the study center.
Sample analysis
Study samples were analyzed by AXYS Analytical Services, of Sidney, British Columbia, using high-resolution gas chromatography/high resolution mass spectrometry. AXYS analyzed an average of 2.5 gram of lipid, for 44 PBDE congeners. Reportable levels of 35 PBDE congeners were detected in the breast milk samples.
Sample preparation and analysis: After addition of surrogate standard solution, the milk samples were liquid/liquid extracted with 2:1 acetone/hexane. The extract was reduced in volume and cleaned up using gel permeation, acid/base silica, Florisil and alumina chromatographic columns. The final extracts were reduced to a volume of 20 uL and spiked with 2 uL of the labeled recovery (internal) standard for a final volume of 22 uL; 1 uL was analyzed.
Analysis was conducted by high resolution gas chromatography/high resolution mass spectrometry (HRGC/HRMS) on an AUTOSPEC ULTIMA high resolution MS equipped with an HP 6890 gas chromatograph, a CTC auto-sampler, and an Alpha data system running Micromass software. A 30 m chromatography column was coupled directly to the MS source.
Detection of trace congeners
Samples were analyzed in 2 batches with 3 laboratory blanks per batch. The reporting limit was definted as 2 times the 99% confidence level for background contamination. The 99% confidence level was calculated as three times the standard deviation of the three background samples, assuming that non-detects were 100% of the detection limit. We included only the reported levels that were >2 times the 99% confidence level when calculating a woman's body burden of PBDEs. All but 3 reported values were more than double the 99% confidence level. These 3 values were omitted from our analysis (PBDE- 71, 206 and 208 in three separate participants.)
Appendix 2: Detailed study results
Results for individual participants (parts per billion PBDEs in breast milk lipid)
|
PBDE congener |
Participant's state of residence |
|||||||||||||||||||
|
CA1 |
CA2 |
CA3 |
CA4 |
CO |
DC |
FL |
GA |
MA |
MA |
MI1 |
MI2 |
MT1 |
MT2 |
MO |
OR |
TN |
TX |
VA |
WA |
|
|
3 |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
0.10 |
ND |
ND |
0.20 |
ND |
ND |
ND |
ND |
|
7 |
ND |
ND |
ND |
ND |
0.01 |
ND |
0.005 |
0.01 |
ND |
ND |
ND |
ND |
0.003 |
ND* |
0.04 |
0.02 |
ND |
0.003 |
ND |
ND |
|
8 + 11 |
ND |
ND* |
0.01 |
ND* |
0.01 |
ND |
0.01 |
0.01 |
ND |
ND |
0.01 |
ND |
0.01 |
0.005 |
0.06 |
0.03 |
ND |
ND* |
ND |
ND |
|
12 |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
0.01 |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
|
13 |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
0.01 |
ND |
ND |
ND |
ND |
|
15 |
0.40 |
1.17 |
1.86 |
0.36 |
0.83 |
0.31 |
0.42 |
0.93 |
0.41 |
0.29 |
0.85 |
0.22 |
0.32 |
0.75 |
17.70 |
1.04 |
0.19 |
0.49 |
0.30 |
0.45 |
|
17 |
0.03 |
0.09 |
0.06 |
0.03 |
0.14 |
0.04 |
0.08 |
0.11 |
0.05 |
0.03 |
0.16 |
0.04 |
0.09 |
0.08 |
0.46 |
0.47 |
0.02 |
0.07 |
0.02 |
0.02 |
|
25 |
0.004 |
0.02 |
0.01 |
0.01 |
0.04 |
0.002 |
0.04 |
0.01 |
0.01 |
0.004 |
0.02 |
0.01 |
0.03 |
0.01 |
0.05 |
0.06 |
ND |
0.01 |
ND* |
0.003 |
|
28 + 33 |
1.26 |
3.38 |
3.14 |
1.23 |
13.70 |
0.93 |
8.06 |
10.20 |
1.66 |
0.89 |
7.67 |
1.02 |
1.73 |
3.09 |
43.80 |
19.10 |
0.35 |
2.05 |
0.36 |
1.12 |
|
35 |
ND* |
ND |
ND |
ND |
ND* |
ND |
ND |
0.02 |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND* |
ND |
ND |
ND |
|
37 |
0.01 |
0.03 |
0.03 |
ND* |
0.08 |
0.01 |
0.08 |
0.06 |
0.02 |
0.01 |
0.06 |
0.01 |
0.02 |
0.03 |
0.40 |
0.18 |
0.01 |
0.02 |
0.01 |
0.01 |
|
47 |
14.60 |
30.80 |
22.20 |
10.80 |
130.00 |
11.60 |
111.00 |
104.00 |
20.40 |
15.70 |
116.00 |
17.90 |
30.70 |
50.10 |
589.00 |
379.00 |
5.53 |
27.40 |
5.56 |
6.51 |
|
49 |
0.14 |
0.40 |
0.21 |
0.12 |
1.00 |
0.11 |
0.29 |
0.52 |
0.15 |
0.08 |
1.19 |
0.08 |
0.38 |
0.47 |
2.20 |
3.69 |
0.04 |
0.21 |
0.04 |
0.06 |
|
66 |
0.14 |
0.31 |
0.24 |
0.13 |
1.16 |
0.09 |
0.98 |
1.27 |
0.15 |
0.14 |
1.72 |
0.12 |
0.37 |
0.94 |
6.05 |
8.37 |
0.06 |
0.29 |
0.06 |
0.07 |
|
71 |
0.01 |
0.03 |
0.02 |
0.01 |
0.05 |
0.01 |
0.04 |
0.04 |
0.01 |
0.01 |
0.13 |
0.01 |
0.05 |
0.06 |
ND* |
0.33 |
ND# |
0.01 |
0.01 |
0.004 |
|
75 |
0.01 |
0.03 |
0.02 |
0.01 |
0.12 |
0.01 |
0.05 |
0.09 |
0.02 |
0.01 |
0.09 |
0.02 |
0.04 |
0.06 |
0.36 |
0.43 |
0.01 |
0.02 |
0.01 |
0.01 |
|
85 |
0.20 |
0.45 |
0.47 |
0.15 |
1.03 |
0.16 |
2.13 |
1.03 |
0.23 |
0.15 |
2.41 |
0.42 |
0.65 |
1.17 |
17.10 |
17.00 |
0.07 |
0.37 |
0.08 |
0.05 |
|
99 |
2.58 |
5.75 |
4.55 |
1.85 |
15.50 |
2.29 |
15.10 |
10.90 |
2.47 |
2.28 |
27.00 |
3.45 |
9.99 |
16.30 |
91.10 |
200.00 |
1.00 |
4.38 |
1.36 |
0.76 |
|
100 |
2.22 |
4.25 |
4.86 |
1.72 |
17.50 |
1.45 |
25.00 |
16.60 |
4.95 |
2.51 |
17.80 |
5.76 |
7.92 |
6.20 |
171.00 |
71.80 |
0.86 |
3.70 |
1.89 |
0.57 |
|
119 |
ND |
ND |
0.02 |
0.01 |
0.05 |
ND |
ND |
ND |
0.07 |
ND |
ND |
ND |
ND |
0.02 |
ND |
0.22 |
ND |
ND* |
ND |
0.004 |
|
126 |
0.01 |
ND |
0.01 |
ND* |
0.02 |
ND* |
ND |
ND* |
0.02 |
0.005 |
0.04 |
0.01 |
ND |
0.02 |
0.22 |
ND |
ND* |
0.01 |
ND |
ND |
|
138 + 166 |
0.03 |
0.05 |
0.06 |
0.02 |
0.12 |
0.02 |
0.27 |
0.11 |
0.04 |
0.02 |
0.27 |
0.08 |
0.09 |
0.11 |
1.76 |
1.66 |
ND* |
0.04 |
0.02 |
ND* |
|
140 |
0.03 |
0.07 |
0.12 |
0.02 |
0.22 |
0.02 |
0.21 |
0.10 |
0.05 |
0.02 |
0.28 |
0.08 |
0.12 |
0.06 |
1.93 |
0.76 |
0.01 |
0.05 |
0.05 |
0.01 |
|
153 |
2.43 |
31.60 |
40.50 |
1.91 |
18.10 |
2.32 |
12.00 |
9.91 |
2.75 |
2.39 |
57.20 |
5.25 |
16.20 |
10.20 |
122.00 |
36.50 |
1.07 |
4.76 |
17.50 |
1.30 |
|
154 |
0.14 |
0.31 |
0.30 |
0.12 |
0.88 |
0.10 |
1.29 |
0.73 |
0.38 |
0.12 |
1.35 |
0.32 |
0.64 |
0.68 |
9.80 |
11.70 |
0.06 |
0.23 |
0.09 |
0.04 |
|
155 |
0.05 |
0.08 |
0.07 |
0.03 |
0.26 |
0.03 |
0.33 |
0.23 |
0.31 |
0.04 |
0.30 |
0.07 |
0.12 |
0.12 |
2.50 |
1.75 |
0.02 |
0.07 |
0.01 |
ND* |
|
183 |
0.12 |
0.08 |
0.08 |
0.07 |
0.26 |
0.05 |
0.12 |
0.31 |
0.18 |
0.10 |
0.09 |
0.14 |
0.21 |
0.09 |
0.46 |
0.31 |
0.06 |
0.09 |
0.07 |
0.07 |
|
190 |
ND |
0.01 |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
ND |
|
206 |
0.01 |
0.01 |
ND* |
ND# |
0.01 |
ND |
0.004 |
ND* |
ND |
ND* |
ND* |
ND* |
0.04 |
ND* |
0.02 |
ND* |
0.01 |
ND* |
0.01 |
ND* |
|
207 |
0.09 |
ND* |
ND* |
0.03 |
0.11 |
0.03 |
0.03 |
0.10 |
ND* |
ND* |
0.08 |
0.04 |
0.13 |
0.04 |
ND* |
0.03 |
0.02 |
0.04 |
0.05 |
0.03 |
|
208 |
ND* |
ND* |
ND* |
ND* |
0.02 |
0.005 |
ND# |
ND* |
ND* |
ND* |
0.01 |
ND* |
0.04 |
0.01 |
ND* |
ND* |
ND* |
0.01 |
ND* |
0.005 |
|
209 |
ND* |
ND* |
ND* |
0.10 |
0.21 |
0.10 |
0.11 |
0.21 |
ND* |
0.08 |
0.25 |
0.13 |
1.23 |
0.17 |
0.40 |
0.20 |
0.13 |
0.13 |
0.30 |
0.13 |
|
Total PBDEs |
24.51 |
78.92 |
78.84 |
18.71 |
201.41 |
19.68 |
177.64 |
157.50 |
34.32 |
24.87 |
234.97 |
35.17 |
71.23 |
90.78 |
1078.41 |
754.84 |
9.50 |
44.45 |
27.77 |
11.22 |
ND= Not detected
ND*= Sample did not meet one or more requirements for quantification
ND# = Less than 2 times the 99% Confidence Interval for background contamination. See Appendix 1
See below for a summary of findings by congener
Congeners not detected in any samples = PBDE 1, 2, 10, 30, 32, 77, 105, 116, 181
Results for individual PBDE congeners (parts per billion in breast millk lipid)
|
PBDE Congener |
Number of bromines |
Class of PBDE |
Percent of samples with detectable levels |
Minimum value |
Median value |
Average value |
Maximum value |
|
3 |
1 |
mono |
10% |
0.1 |
0.15 |
0.15 |
0.2 |
|
7 |
2 |
di |
35% |
0.003 |
0.01 |
0.01 |
0.04 |
|
8 + 11 |
2 |
di |
45% |
0.005 |
0.01 |
0.02 |
0.06 |
|
12 |
2 |
di |
5% |
0.01 |
0.01 |
0.01 |
0.01 |
|
13 |
2 |
di |
5% |
0.01 |
0.01 |
0.01 |
0.01 |
|
15 |
2 |
di |
100% |
0.19 |
0.43 |
1.46 |
17.7 |
|
17 |
3 |
tri |
100% |
0.02 |
0.06 |
0.1 |
0.47 |
|
25 |
3 |
tri |
90% |
0.002 |
0.01 |
0.02 |
0.06 |
|
28 + 33 |
3 |
tri |
100% |
0.35 |
1.89 |
6.24 |
43.8 |
|
35 |
3 |
tri |
5% |
0.02 |
0.02 |
0.02 |
0.02 |
|
37 |
3 |
tri |
95% |
0.01 |
0.02 |
0.06 |
0.4 |
|
47 |
4 |
tetra |
100% |
5.53 |
24.8 |
84.94 |
589 |
|
49 |
4 |
tetra |
100% |
0.04 |
0.21 |
0.57 |
3.69 |
|
66 |
4 |
tetra |
100% |
0.06 |
0.26 |
1.13 |
8.37 |
|
71 |
4 |
tetra |
90% |
0.004 |
0.02 |
0.05 |
0.33 |
|
75 |
4 |
tetra |
100% |
0.01 |
0.02 |
0.07 |
0.43 |
|
85 |
5 |
penta |
100% |
0.05 |
0.43 |
2.27 |
17.1 |
|
99 |
5 |
penta |
100% |
0.76 |
4.47 |
20.93 |
200 |
|
100 |
5 |
penta |
100% |
0.57 |
4.91 |
18.43 |
171 |
|
119 |
5 |
penta |
35% |
0.004 |
0.02 |
0.06 |
0.22 |
|
126 |
5 |
penta |
50% |
0.005 |
0.01 |
0.04 |
0.22 |
|
138 + 166 |
6 |
hexa |
90% |
0.02 |
0.07 |
0.26 |
1.76 |
|
140 |
6 |
hexa |
100% |
0.01 |
0.07 |
0.21 |
1.93 |
|
153 |
6 |
hexa |
100% |
1.07 |
10.06 |
19.79 |
122 |
|
154 |
6 |
hexa |
100% |
0.04 |
0.32 |
1.46 |
11.7 |
|
155 |
6 |
hexa |
95% |
0.01 |
0.08 |
0.33 |
2.5 |
|
183 |
7 |
hepta |
100% |
0.05 |
0.09 |
0.15 |
0.46 |
|
190 |
7 |
hepta |
5% |
0.01 |
0.01 |
0.01 |
0.01 |
|
206 |
9 |
nona |
40% |
0.004 |
0.01 |
0.01 |
0.04 |
|
207 |
9 |
nona |
75% |
0.02 |
0.04 |
0.06 |
0.13 |
|
208 |
9 |
nona |
35% |
0.005 |
0.01 |
0.01 |
0.04 |
|
209 |
10 |
deca |
80% |
0.08 |
0.15 |
0.24 |
1.23 |
Number of congeners looked for = 44
Number of congeners detected = 35
Not detected = PBDE 1, 2, 10, 30, 32, 77, 105, 116, 181
Appendix 3: Participant info/photos
Twenty first-time mothers participated in this study. Each collected a breast milk sample between 7 and 100 days postpartum, with an average of 44 days. Ten of the 20 participants collected breast milk samples through hand expression. Ten participants expressed milk directly into the study jars using commercially available breast pumps.
The participants reside in 14 states, representing over a quarter of the United States including: California (4), Colorado, the District of Columbia, Florida, Georgia, Massachusetts (2), Missouri, Michigan (2), Montana (2), Oregon, Tennessee, Texas, Virginia, and Washington. Nineteen of the 20 study participants have spent the majority of their lives living in the US.
The majority of the study participants (18 of 20) are Caucasian; one woman is Vietnamese and another reported multiple races. The participant's ages ranged from 29 - 40 years of age with an average age of 33 years. Nineteen of 20 participants are college graduates. The women who participated in the study had a pre-pregnancy Body Mass Index (BMI) range from 19.5 to 34.7 kg/m2 with a median BMI of 21.1 kg/m2. None of the participants were current smokers. Five women were former smokers.
The study participants report eating very little fast food. Several participants consumed little or no meat, fish or seafood. Eight participants reported that they eat three or fewer servings per week, though all but one consume diary products.
None of the women report atypical exposures to foam or plastic products in their homes, neighborhoods or workplaces that might contain PBDEs. Two participants reported that they had reupholstered furniture in their homes in the past 3 years. More than half of the women worked in occupations that required several hours of work per day in front of a computer. Three women report very little computer use.
Study Participants
 Rani and Samuel |
 Teri and Natalie |
|
 Angie and Graceanna |
 Darcy and Katelyn |
|
 Erika and Hannelore |
 Jennifer and Elijah |
|
 Katrina and Ruby |
 Laurie and Connor |
|
 Jenna and Stella |
 Leila and Max |
|
 Margaret and Tavin |
 Susan and Luke |
|
 Suzanne and Mckenzie |
 Tiffany and Samantha |
Acknowledgements
Principal authors: Sonya Lunder and Renee Sharp
Research assistance: Amy Ling and Caroline Colesworthy
Editors: Jane Houlihan, Bill Walker and Richard Wiles
Website, design and graphics: T.C. Greenleaf
Special thanks to each of the 20 women who volunteered to participate in this study. The donation of your time and breast milk will help foster change, starting with better decisions on the part of the government and industry in a transition to safer chemical fire retardants, and with better-informed consumer choices on the part of families.
We are grateful for the support of Dr. Angela Falany and Dr. Michael Litrel of Cherokee Women's OB/GYN in Cherokee County, Ga. Thanks to Dr. Tom McDonald of California EPA, Dr. Linda Birnbaum of the U.S. EPA, Dr. Arnold Schecter of the University of Texas, Dr. Kim Hooper of the California EPA Hazardous Materials Lab, and Ruthann Rudel of Silent Spring Institute for advice on our research or for reviewing the technical report. Thanks also to Public Research Works in Texas, and Women's Voices for the Earth in Montana, for recruiting participants and assisting with release of the report.
This report was made possible by grants from the Richard & Rhoda Goldman Fund, the Beldon Fund, the John Merck Fund and the California Wellness Foundation. The opinions expressed in this report are those of the authors and editors and do not necessarily reflect the views of our supporters. EWG is responsible for any errors of fact or interpretation contained in this report.